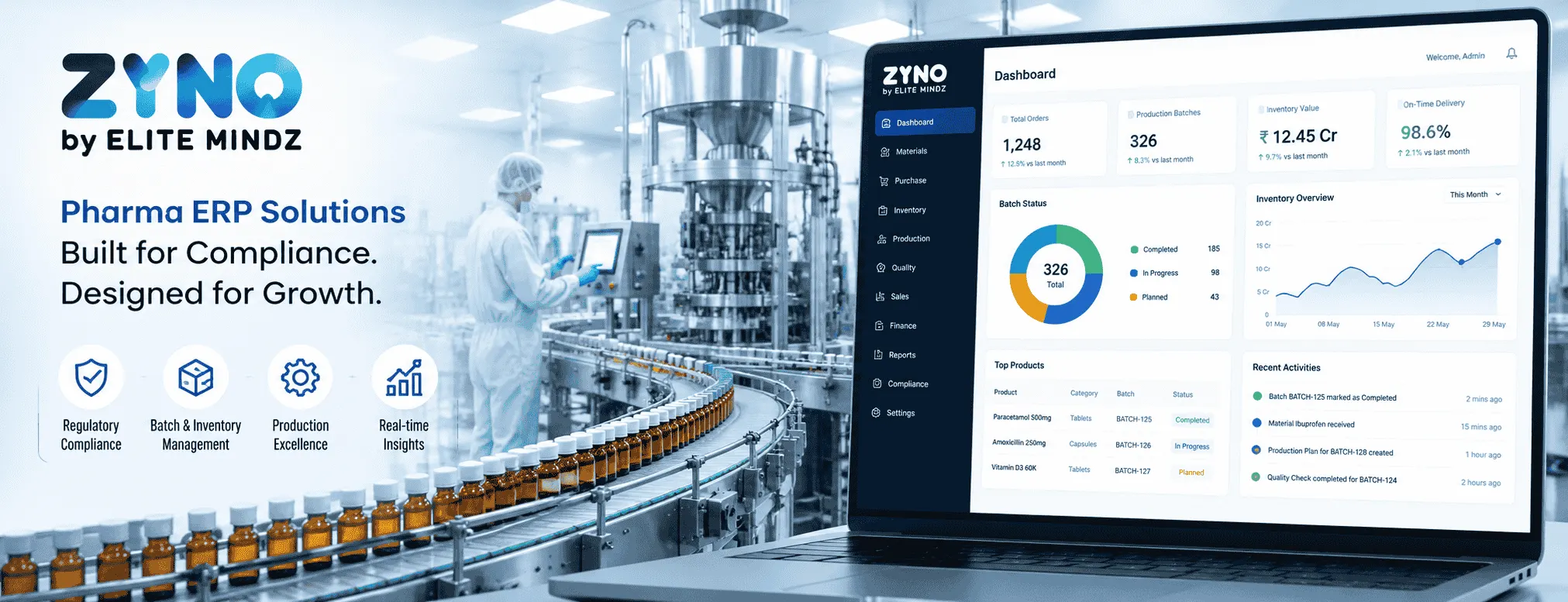
×
×

India is the pharmacy of the world — and that title comes with enormous responsibility. With over 3,000 pharma companies and 10,500 manufacturing units spread across the country, the Indian pharmaceutical industry is growing at breakneck speed. The market, valued at USD 57.6 billion in 2026, is projected to reach USD 120–130 billion by 2030 — growing at a CAGR of over 8%.
In FY25 alone, the sector recorded an annual turnover of ₹4.72 lakh crore, with pharmaceutical exports hitting USD 30.38 billion — shipped to over 191 countries. India now ranks 3rd globally in pharmaceutical production by volume and is the world's largest supplier of generic medicines, accounting for 20% of global generic supply.
But with this scale comes complexity: regulatory audits, batch traceability, GST compliance, expiry management, and multi-plant operations — all happening simultaneously. Managing all of this on spreadsheets or disconnected legacy systems is no longer viable — and regulators are making that clearer every year.
That is precisely why pharma ERP software has become a non-negotiable investment for pharmaceutical manufacturers, distributors, and API companies in India in 2026.
In this guide, we cover everything you need to know before selecting a pharma ERP — from must-have features and India-specific compliance requirements to implementation tips and common mistakes to avoid.
A Pharma ERP (Enterprise Resource Planning) software is an industry-specific business management system designed to streamline and integrate the operations of pharmaceutical companies — from raw material procurement and batch manufacturing to quality control, compliance documentation, inventory management, and financial reporting.
Unlike generic ERP systems, pharmaceutical ERP software is purpose-built to handle the unique demands of a regulated industry. It manages complex workflows such as:
In simple terms, pharma ERP is the central nervous system of your pharmaceutical operation — connecting every department, every workflow, and every compliance requirement into one unified platform.
India's pharmaceutical sector has grown into a global powerhouse. Here is why the pressure on operations has never been higher — and why 2026 is a turning point:
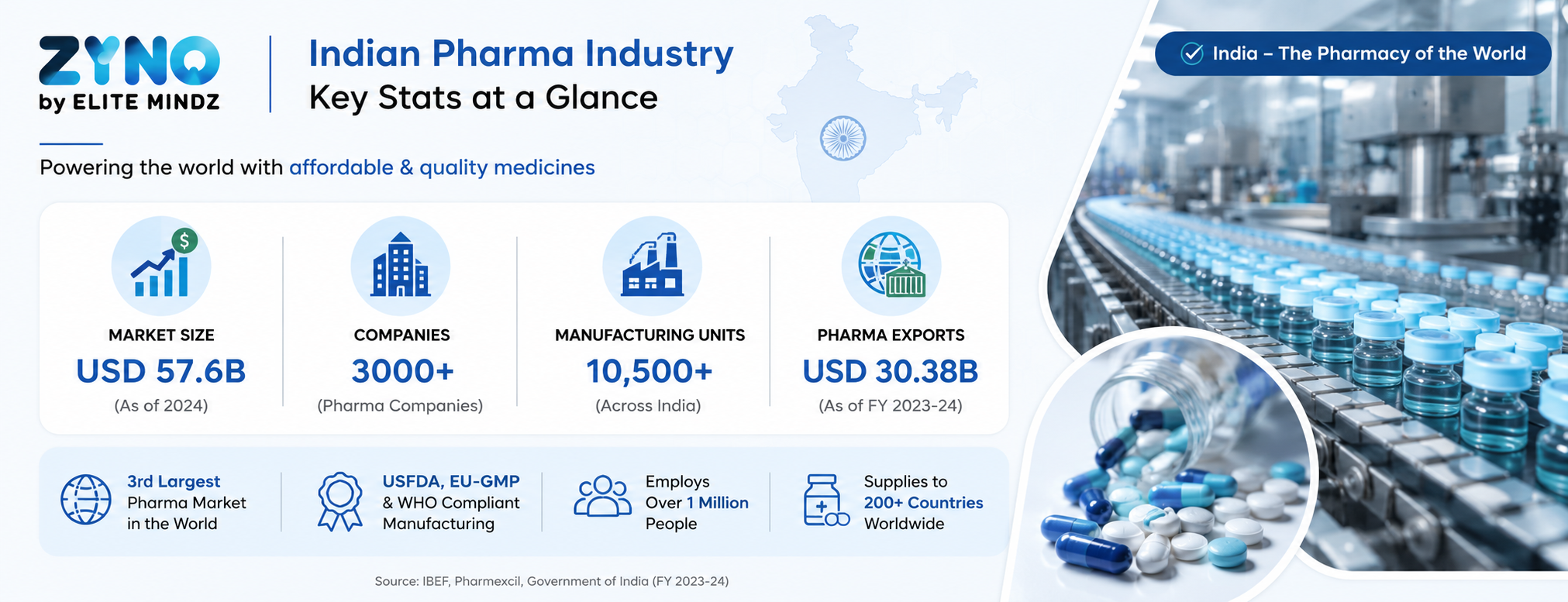
India currently ranks 3rd globally by pharmaceutical production volume and supplies over 20% of the world's generic medicines. As of 2026, the country has more than 2,000 WHO-GMP approved manufacturing units and over 670 USFDA-approved facilities — the highest number outside the United States. The government's PLI (Production Linked Incentive) scheme, with a total outlay of ₹15,000 crore running through 2028–29, is accelerating domestic API manufacturing and attracting new investment — adding further operational complexity for manufacturers.
Managing operations at this scale requires automation, traceability, and real-time data — things no spreadsheet or legacy system can reliably provide.
The Central Drugs Standard Control Organisation (CDSCO), India's primary drug regulatory authority, has significantly tightened enforcement. In 2025 alone, 18% of inspected manufacturing sites failed to meet updated data integrity benchmarks. In 2026, stricter sterility and data-integrity rules took effect, and non-compliant facilities face plant shutdowns, regulatory action, and loss of export certifications. As enforcement escalates through 2027, audit-ready documentation is no longer optional.
India's GST framework continues to evolve. Pharmaceutical companies must generate IRN-linked e-invoices, manage HSN-code-based journal entries, file GST returns, and generate e-Way bills for every interstate shipment. The government's push for real-time tax reporting makes manual processing a liability. A pharma ERP eliminates this risk with automated fiscal compliance built in.
India's pharmaceutical exports reached USD 30.38 billion in FY25, growing at a CAGR of 7% over the past decade, with exports to 191 countries — 50% of which go to highly regulated markets such as the US and EU. Companies exporting to these markets must comply with FDA 21 CFR Part 11 and EU-GMP standards — requirements that demand digital audit trails, electronic signatures, and validated systems. In 2026, India also concluded a Free Trade Agreement with New Zealand providing zero-duty access for pharma exports, opening new markets that carry their own compliance requirements. Only a purpose-built pharma ERP can reliably meet all of this.
One of the most common mistakes Indian pharma companies make is trying to adapt a generic ERP — built for retail, manufacturing, or trading — to the highly regulated world of pharmaceuticals. The result is always the same: expensive customizations, compliance gaps, and audit failures.
Here is a detailed, side-by-side comparison of what generic ERP provides versus what a purpose-built pharma ERP delivers:
| Feature / Capability | Generic ERP | Pharma ERP |
|---|---|---|
| Batch Manufacturing Records (BMR) | Not Available | Auto-generated, e-signed, version-controlled |
| Master Formula / Recipe Management | Basic BOM only | Version-controlled MFR with potency & yield tracking |
| Batch Traceability | Not Available | Bi-directional lot traceability end-to-end |
| Expiry Date & Shelf-Life Management | Basic expiry alerts only | FEFO logic, auto-blocking of expired/quarantined stock |
| Quality Control (QC) Workflows | Not Available | In-process testing, OOS/OOT investigation, COA generation |
| CAPA Management | Not Available | Structured deviation, non-conformance & CAPA workflows |
| CDSCO / Schedule M Compliance | Not Available | Built-in compliance templates and documentation |
| FDA 21 CFR Part 11 Audit Trail | Not Available | Unalterable, time-stamped audit trails |
| Electronic Signatures | Not Available | Role-based e-signatures at critical checkpoints |
| WHO-GMP Documentation | Not Available | SOP management, deviation tracking, validation records |
| Document Management System (DMS) | Basic document storage | Version-controlled approval workflow DMS |
| GST & E-Invoicing (India) | Varies by vendor | Native GST, IRN e-invoice, e-Way bill, TDS/TCS |
| Schedule H / Narcotic Drug Compliance | Not Available | Prescription tracking and statutory reports |
| Yield Calculation & Loss Tracking | Not Available | Actual vs expected yield analysis |
| Stability Testing Management | Not Available | Study scheduling and result logging |
| Equipment Calibration & Maintenance | Basic asset tracking | Calibration scheduling and GMP logs |
| Multi-Plant / Multi-Warehouse Operations | Limited support | Centralized control across all locations |
| PCD / C&F / Stockist Distribution | Not Available | Channel pricing, schemes, and credit limits |
| Regulatory Reporting | Not Available | Audit-ready compliance reports |
| System Validation (IQ/OQ/PQ) | Not Supported | Fully supported with documentation |
| Cold Chain Monitoring | Not Available | Temperature-sensitive tracking and alerts |
| Label & Packaging Compliance | Generic label printing | Drug-specific labelling and serialization |
Verdict: A generic ERP can handle your finance, HR, and basic inventory. But the moment you need CDSCO audit trails, batch manufacturing records, quality workflows, or FDA compliance — a generic ERP falls short. Trying to customize it fills that gap partially, at enormous cost, and still leaves regulatory blind spots. For pharma, the only viable path is a purpose-built pharma ERP.
Before investing in pharma ERP software, it helps to understand exactly what problems it solves. Here are the most common pain points Indian pharma businesses face — and how ERP addresses them:

Paper-based or Excel-based Batch Manufacturing Records are prone to errors, hard to audit, and difficult to retrieve during inspections. Pharma ERP automates BMR generation, links every batch to its raw materials, equipment, and quality tests, and stores records with version control and electronic signatures.
Managing thousands of raw material lots with different expiry dates — especially with FEFO (First Expiry, First Out) logic — is nearly impossible manually. ERP automatically blocks expired or quarantined stock and sends alerts before materials hit critical dates.
CDSCO inspections, GMP audits, WHO-GMP certifications, and FDA site visits all require mountains of documentation. A pharma ERP maintains SOPs, audit trails, CAPA records, and deviation logs in a structured, instantly retrievable format so your team is always audit-ready.
When QC and production operate in separate systems, data gaps lead to batch release delays, missed non-conformances, and compliance risks. ERP integrates QC, QA, and production into a single workflow, ensuring quality data flows automatically to the right teams.
Without real-time inventory visibility, pharma companies face either excess stock (with write-off risk) or stockouts (with production delays). ERP provides live stock dashboards, auto-reorder triggers, and batch-wise inventory tracking across multiple warehouses.
Manual GST calculations across product categories, tax slabs, and interstate transactions lead to filing errors and penalties. ERP automates GST computation, e-invoice generation, e-Way bill creation, TDS/TCS management, and financial reporting.
Not all ERP systems are built for pharmaceuticals. When evaluating pharma ERP software, ensure it includes these industry-critical features:

The ERP must support process manufacturing — where a product is the result of a precise formula, not just an assembly of parts. Look for strict version control on Master Formula Records, automated potency adjustments, yield calculation, and batch scaling capabilities.
Auto-generation of BMR and Batch Packaging Records (BPR) with controlled access, real-time data capture, and e-signature workflows eliminates paper trails and reduces audit preparation time from weeks to hours.
An in-built QMS should handle deviation management, Out-of-Specification (OOS) and Out-of-Trend (OOT) investigations, CAPA workflows, Certificate of Analysis (COA) generation, and stability testing — all linked directly to production batches.
Pharmaceutical inventory must move on a First Expiry, First Out basis. The ERP must automatically suggest FEFO-compliant lot selection during material issuance, track cold chain requirements, and block expired stock from being used in production.
The system must generate time-stamped, unalterable audit trails for every data entry and approval. Electronic signatures must be compliant with 21 CFR Part 11 (for exporters) and support CDSCO and Schedule M documentation requirements.
For Indian pharma businesses, native GST integration is non-negotiable. The ERP must automate tax calculation, generate IRN-linked e-invoices, create e-Way bills, manage TDS/TCS deductions, and support HSN-code-based accounting.
Large Indian pharma companies often operate across multiple manufacturing sites and distribution warehouses. The ERP must provide centralized visibility, transfer management, and consolidated reporting across all locations.
Procurement in pharma requires approved vendor lists, material test approvals, purchase order workflows, and GRN quality inspection before stock is accepted. ERP should enforce these steps automatically.
Indian pharma distribution includes complex models — C&F agents, stockists, PCD (Propaganda Cum Distribution) franchises, and direct hospital sales. The ERP must handle multiple pricing tiers, scheme management, credit limits, and route-to-market tracking.
All SOPs, validation documents, regulatory submissions, and training records should be stored in a centralized, version-controlled DMS within the ERP — not in physical folders or email threads.
This is the section that separates a generic ERP from a true pharmaceutical ERP for India. The regulatory landscape here is multi-layered:
The Central Drugs Standard Control Organisation (CDSCO) mandates compliance with Schedule M of the Drugs and Cosmetics Act, 1940 — which defines Good Manufacturing Practices for pharmaceutical manufacturers in India. Your ERP must:
With over 2,000 WHO-GMP approved manufacturing units in India, maintaining this certification is critical for exports to emerging markets. ERP must support WHO-GMP documentation protocols including batch records, deviation handling, and out-of-specification management.
Indian companies exporting to the United States — and India has over 670 USFDA-approved facilities — must comply with 21 CFR Part 11. This requires the ERP to:
All pharmaceutical transactions in India are subject to GST. The ERP must natively support:
Pharma ERP for Indian distributors and retail chains must handle Schedule H and Schedule H1 drug restrictions — tracking prescription-only sales, maintaining purchase registers, and generating statutory reports required by the licensing authority.
Implementing the right ERP system delivers measurable outcomes across your business:
Faster Batch Release — Automated QC workflows and digital BMRs eliminate manual handoffs, reducing batch release time by up to 40-60% in most implementations.
Audit Readiness, Always — Real-time documentation and audit trails mean your team is never scrambling before an inspection. CDSCO audits and FDA site visits become routine rather than stressful.
Reduced Inventory Write-offs — FEFO logic, expiry alerts, and real-time stock visibility significantly reduce losses from expired or overstocked raw materials and finished goods.
Improved Supply Chain Visibility — From purchase order to product dispatch, every step is tracked and visible. This directly reduces production delays caused by material shortages or supplier defaults.
GST Compliance Without Manual Effort — Automated tax computation and e-invoicing eliminate GST filing errors and reduce the manpower burden of financial compliance.
Scalability for Growth — Whether you add a new manufacturing plant, a new product line, or a new distribution territory, a well-implemented ERP scales with your business without requiring system overhauls.
Data-Driven Decision Making — Live dashboards, production analytics, and financial reports give management real-time visibility to make informed decisions on pricing, procurement, and production planning.
With dozens of options in the Indian market, choosing the right pharma ERP can be overwhelming. Use this framework to evaluate your options:
Are you a formulation manufacturer, API producer, nutraceutical company, CDMO, or pharma distributor? Your business type determines which modules are most critical. A formulation manufacturer needs strong BMR automation; a distributor needs channel management and GST compliance.
List the regulatory bodies you are currently certified by or plan to be. CDSCO/Schedule M compliance is mandatory for all. Add WHO-GMP, FDA 21 CFR Part 11, or EU-GMP requirements if you export to regulated markets.
Ensure your ERP vendor understands India's pharma regulatory landscape — not just generic international standards. Native GST integration, Schedule M documentation, and CDSCO audit trail requirements must be built into the system, not bolted on.
Consider your IT infrastructure, data security requirements, and budget. Cloud-based pharma ERP typically offers lower upfront costs, automatic compliance updates, and remote access. On-premise suits companies with strict data sovereignty needs or limited internet reliability at manufacturing sites.
Your ERP should integrate seamlessly with existing systems — LIMS (Lab Information Management Systems), MES (Manufacturing Execution Systems), financial software, and government portals for e-invoicing and GST filing.
Request references from companies in your specific segment — tablet manufacturers, injectable producers, API companies, etc. Generic ERP implementation experience does not qualify a vendor for pharma.
Look beyond the license fee. Factor in implementation costs, training, annual maintenance, compliance update charges, and scalability costs as your business grows.
This is one of the most common questions Indian pharma companies ask when evaluating ERP options.
Cloud-Based Pharma ERP is increasingly the preferred choice for Indian manufacturers for several reasons. Automatic regulatory patches mean your system stays current with the latest CDSCO, GST, and e-invoicing rules without manual updates. Lower Total Cost of Ownership (TCO) removes the need for expensive on-site server infrastructure. Remote access allows management teams, quality officers, and sales teams to work from anywhere.
On-Premise ERP remains relevant for large enterprises with dedicated IT departments, strict data sovereignty requirements, or manufacturing sites in areas with unreliable internet connectivity. However, the upfront capital investment and ongoing IT maintenance costs are significantly higher.
For most small to mid-sized Indian pharma manufacturers and distributors, a cloud-based pharma ERP with strong data security, regular compliance updates, and local support is the smarter long-term investment.
A successful pharma ERP implementation in India typically follows this roadmap:
Phase 1 — Business Process Mapping (4-6 weeks)
Document your existing workflows across production, QC, inventory, procurement, and finance. Identify gaps, pain points, and compliance requirements. This phase is the foundation of everything that follows.
Phase 2 — System Configuration (6-8 weeks)
Configure the ERP to match your business processes — product master setup, formula management, approval workflows, user roles, and compliance templates for CDSCO and GMP documentation.
Phase 3 — Data Migration (2-4 weeks)
Migrate historical data including customer master, vendor master, inventory records, and product formulations. Data accuracy at this stage is critical to avoid downstream errors.
Phase 4 — Training (2-4 weeks)
Train all users — from production floor operators and QC analysts to finance teams and management. Role-based training ensures each team member understands their specific workflows within the system.
Phase 5 — Validation and UAT (2-4 weeks)
For companies operating under FDA or WHO-GMP guidelines, system validation (IQ, OQ, PQ) is mandatory. User Acceptance Testing (UAT) ensures all configured workflows match real business requirements before go-live.
Phase 6 — Go-Live and Hyper-Care Support (4-8 weeks)
Launch in a phased manner — starting with one plant or one department — before rolling out company-wide. Post-go-live support from your vendor is critical during this period to resolve issues quickly.
Learning from others' mistakes saves time, money, and compliance headaches. Here are the most common errors Indian pharma companies make when selecting and implementing ERP:
Choosing a Generic ERP and Customizing It for Pharma — Customizing a standard ERP to meet pharma requirements is expensive, time-consuming, and often creates compliance gaps. Always prefer a purpose-built pharma ERP.
Underestimating Implementation Complexity — ERP implementation in pharma is not a plug-and-play process. It requires deep involvement from your quality, production, and finance teams — and a realistic timeline of 6-12 months for a full deployment.
Ignoring Post-Go-Live Support — Many companies focus entirely on the software purchase price and neglect to evaluate the vendor's after-sales support. In a regulated industry, a compliance update or system issue needs to be resolved in hours, not weeks.
Not Involving QC and Regulatory Teams Early — Your quality and compliance teams must be involved from day one of ERP selection. They understand the regulatory requirements your system must meet — and discovering gaps after go-live is extremely costly.
Skipping System Validation — For companies in regulated markets, skipping IQ/OQ/PQ validation is not an option. It exposes you to data integrity warnings and 483 observations during FDA inspections.
Choosing the Cheapest Option — Pharma ERP is a strategic investment in your compliance infrastructure. Selecting the lowest-cost option without evaluating features and support quality often results in a system that cannot scale or meet evolving regulatory requirements.
What is pharma ERP software?
Pharma ERP software is an enterprise resource planning system specifically designed for pharmaceutical companies. It integrates production, quality control, inventory, compliance, procurement, and finance into one platform, helping pharma businesses manage complex regulatory requirements like GMP, CDSCO, and FDA standards while improving operational efficiency.
Is pharma ERP software mandatory in India?
While pharma ERP is not legally mandated by a single regulation, it is effectively required to comply with CDSCO Schedule M documentation standards, GST e-invoicing mandates, and audit trail requirements for regulated market exports. Companies that continue operating without ERP face growing risks of regulatory non-compliance and audit failures.
What is the difference between pharma ERP and general ERP?
A general ERP handles standard business processes like finance, HR, and inventory. A pharma ERP adds industry-specific capabilities including batch manufacturing records, formula management, quality management with CAPA workflows, compliance documentation for GMP and CDSCO, expiry management with FEFO logic, and regulatory audit trails — none of which are available in off-the-shelf generic ERP systems.
Which compliance standards should pharma ERP support in India?
At minimum, pharma ERP for Indian manufacturers should support CDSCO Schedule M (GMP), WHO-GMP documentation, GST and e-invoicing compliance, and drug licensing requirements. Companies exporting to the US must additionally comply with FDA 21 CFR Part 11, while EU exporters must meet EU-GMP standards.
How long does pharma ERP implementation take in India?
Pharma ERP implementation typically takes 6 to 12 months depending on the size of the organization, number of plants, complexity of product portfolio, and the extent of system validation required. Smaller pharma distributors may complete implementation in 3-4 months, while large multi-plant manufacturers with export compliance requirements should plan for 9-12 months.
Can small pharma companies in India afford ERP software?
Yes. Cloud-based pharma ERP solutions have significantly lowered the cost of entry for small and mid-sized pharmaceutical companies in India. Many vendors offer modular pricing, allowing companies to start with core modules like production and inventory and add quality and compliance modules as they grow.
Does pharma ERP help with GST compliance in India?
Yes. A good pharma ERP for India includes native GST support — automating tax calculation, generating e-invoices with IRN, creating e-Way bills, managing TDS/TCS, and enabling GST return filing. This eliminates manual GST processing and reduces the risk of filing errors and penalties.
What is BMR in pharma ERP?
BMR stands for Batch Manufacturing Record — a complete document that records every step taken during the manufacture of a specific batch of a pharmaceutical product. In pharma ERP, BMRs are auto-generated digitally, linked to the batch, signed electronically, and stored with full version control, making them instantly retrievable during audits.
The Indian pharmaceutical industry is at a defining inflection point in 2026. With a market growing toward USD 130 billion by 2030, over 10,500 manufacturing units operating under increasingly strict regulatory oversight, exports of USD 30+ billion reaching 191 countries, and a government PLI scheme pushing domestic API manufacturing to new levels — the operational and compliance demands on Indian pharma companies have never been greater.
The question is no longer whether your pharma company needs an ERP — it is which one and when.
The right pharma ERP software does not just digitize your processes. It becomes the compliance backbone of your operation — ensuring every batch is traceable, every document is audit-ready, every GST transaction is accurate, and every quality event is handled systematically. As the comparison table in this guide makes clear, a generic ERP simply cannot deliver what the pharmaceutical industry requires.
When evaluating your options, prioritize India-specific compliance coverage (CDSCO, Schedule M, GST, e-invoicing), purpose-built pharma functionality (BMR, QMS, FEFO, CAPA), and a vendor with deep implementation experience in your business segment. A well-implemented pharma ERP pays for itself through reduced compliance risk, fewer inventory write-offs, faster batch release, and the operational confidence to scale without chaos.
Looking for a pharma ERP solution built for Indian pharmaceutical manufacturers? Explore how Elite Mindz ZYNO ERP helps pharma companies streamline operations, ensure CDSCO and GMP compliance, and scale with confidence.
If your pharma business is dealing with compliance pressure, batch traceability challenges, GST complexity, or the need to scale operations without adding manual overhead — it may be the right time to evaluate a purpose-built solution.
ZYNO ERP by Elite Mindz is a cloud-based manufacturing and compliance platform built for Indian pharmaceutical companies. It covers the core requirements this guide outlines: batch manufacturing, quality workflows, CDSCO-aligned documentation, native GST compliance, and modular scalability.
Explore ZYNO ERP - Book a free demo to see how ZYNO handles your specific pharma compliance requirements.
(1).webp)
Sneha Singh
Content Writer
Sneha Singh is a B2B tech content strategist with 4+ years of experience. She specializes in SEO-driven SaaS content, whitepapers, and platform-native social media campaigns that simplify complex technology and drive business growth. Sneha's core exp

What is ERP? A Beginner's Guide to Enterprise Resource Planning
Read More →.jpg)
Cloud ERP vs. On-Premise vs. Hybrid: Best ERP Model for 2025
Read More →.jpg)
How To Customize ERP Software Without Breaking The Bank?
Read More →
The Future of Manufacturing ERP in 2026 & Beyond
Read More →.webp)
Best ERP Software in India: 2026 Edition
Read More →
AI in Manufacturing ERP: The Future of Smart Factories
Read More →.jpg)
How ERP Transforms Inventory Management and Reduces Costs
Read More →.jpg)
Top Features to Look for in Inventory Management Software in 2025
Read More →.jpg)
Top 5 Ways Asset Management Software Can Save Your Business Money
Read More →
Why Smart Businesses Are Switching to Vendor Management Systems in 2025?
Read More →.jpg)
Why Manufacturing Companies Need ERP Software
Read More →
Cloud ERP vs. Traditional ERP: Which is Right for You?
Read More →
ERP for Small Business: Is It Worth the Investment?
Read More →
How to Calculate ERP ROI and Why It's Worth the Investment
Read More →
Top 10 ERP Software Companies in India 2026 Updated List (Complete Guide)
Read More →
ERP Implementation Mistakes to Avoid in 2025
Read More →
Cloud ERP vs Edge Computing: What's Right for Modern Manufacturing?
Read More →
The Future of ERP Software and the Role of Generative AI
Read More →
Digital Twin Technology & ERP: Real-Time Insights for Smarter Manufacturing
Read More →
Scalable ERP Solutions Designed for Modern Enterprises
Read More →.jpg)
ERP and IoT Integration: Smart Factory Vision
Read More →.jpg)
Best ERP Software for Manufacturing Businesses in India
Read More →(1).webp)
Top 10 Best Manufacturing ERP Software in India (2026)
Read More →.jpg)
Best ERP Software for Manufacturing Industries in Ethiopia (2026)
Read More →.jpg)
Best School ERP Software Providers in UAE (2025 Edition)
Read More →
Best School ERP Software for 2025: Guide for Educational Institutions
Read More →
Top 10 ERP Software Solutions for Nigerian Manufacturers (2025 Edition)
Read More →
Best ERP Systems for Scalable Retail and POS Integration
Read More →
Top 10 Cloud-Based Manufacturing ERP Solutions in Africa (2025 Edition)
Read More →
Top 10 Cloud-Based Manufacturing ERP in UAE
Read More →
Top 10 Cloud-Based Manufacturing ERP Software in Nigeria (2026 Edition)
Read More →.webp)
Top 10 School ERP Software Companies in Kenya (2025)
Read More →.jpg)
Best School ERP Software Providers in Nigeria (2025)
Read More →.jpg)
Top Cloud-Based Manufacturing ERP in Zimbabwe (2025)
Read More →.jpg)
Best ERP Software in U.K
Read More →.jpg)
Top 10 ERP Software Developers in the U.K. (2025)
Read More →.jpg)
Best ERP Software Providers in France (2025)
Read More →.jpg)
Best ERP Software Companies in Germany : A Comprehensive Guide
Read More →.jpg)
Top 10 ERP Software Development Companies in Qatar
Read More →
10 Best ERP Providers in Germany - 2026 Updated List
Read More →.jpg)
Best ERP Software Firms in Saudi Arabia (2025)
Read More →
Best ERP Service Providers in Indonesia for SMEs and Large Enterprises
Read More →
Top 10 Cloud-Based Manufacturing ERP Systems in Egypt for Modern Factories
Read More →
Top ERP Solutions in the USA 2025: Streamline Your Business with ZYNO ERP
Read More →
Top ERP Solutions in Qatar for Businesses of All Sizes
Read More →
Top Manufacturing ERP Software in India 2026 ZYNO Manufacturing ERP
Read More →
How Manufacturing Companies Reduce Production Delays Using ERP Automation
Read More →.webp)
Why Legal Firms Need ERP to Manage Cases, Billing & Compliance
Read More →.webp)
What Is ERP And How Does It Work
Read More →
How ERP Systems Are Evolving in 2026: What Businesses Need to Know
Read More →
Best ERP Software in India: 2025 Edition
Read More →
Top 10 ERP Software Companies in Delhi NCR
Read More →
How Manufacturing ERP Software Improves Demand Forecasting
Read More →
Top 10 Best School Management Software in India
Read More →.webp)
Manufacturing & Wholesale ERP in Qatar: Complete Industry Guide 2026
Read More →.webp)
Cloud ERP Software in Qatar: Why Businesses Are Moving in 2026
Read More →.webp)
Is ERP a Procurement Software? Key Differences, Benefits & What to Choose
Read More →.webp)
What Is Production Planning and Control? Definition, Objectives & Functions
Read More →
Garment Inventory Management Software: Complete Guide 2026
Read More →
How ERP Reduces Fabric Wastage in Garment Factories
Read More →
Why the Garment Industry Needs a Specialized ERP
Read More →
Features to Look for in Garment Manufacturing ERP Software
Read More →
Best ERP Software for Small Garment Manufacturing Business in India
Read More →
The Real Problems Holding Back Small Garment Manufacturers in India
Read More →.webp)
Garment Manufacturing ERP Software in India: Best Options, Features & Complete Guide 2026
Read More →
Which ERP is Best for Manufacturers in 2026?
Read More →
Distributor & Consumer Management System: The Complete Guide for 2026
Read More →
Best Pharma ERP Software in India 2026 – Features, Compliance & Buying Guide
Read More →
SAP Alternative for Manufacturing in India (2026): 6 Options Compared Honestly
Read More →Partner with ZYNO by Elite Mindz to revolutionize how your business works. Fill out the form, and we’ll reach out with AI-powered solutions made just for you.

We use browser cookies to ensure you get the best experience on our website. Learn more
The form was submitted successfully!
ZYNO Digital
Fueling Smarter Digital Growth
ZYNO Digital fuels smarter digital growth through data-driven marketing, creative storytelling, and performance strategies that boost visibility, engagement, and measurable business outcomes across global platforms.

ZYNO Tech
Engineering the Future of IT
ZYNO Tech delivers cutting-edge IT solutions — including web, mobile, and digital transformation services — helping businesses scale efficiently, innovate faster, and stay ahead in a connected world.

ZYNO AI
AI Built for Business Growth
ZYNO AI harnesses the power of artificial intelligence to automate processes, enhance decision-making, and deliver predictive insights — driving innovation, agility, and measurable business growth.

ZYNO by Elite Mindz
Your Partner in the AI-Powered Future
ZYNO by Elite Mindz offers AI-powered products and services that unify operations, boost productivity, and drive digital transformation — helping businesses scale smarter and innovate faster.
* In just 2 mins you'll get a response
* Your idea is 100% protected by our Non-Disclosure Agreement